
It is thus possible to identify regions of space around the nucleus where there is high probability of locating an electron associated with a specific energy. However, the square of the wave function ,that is, Ψ2 gives the probabilityof an electron of a given energy E, from place to place in a given region around the nucleus. The wave function Ψ in Schrodinger wave equation, has no physical significance except than it represents the amplitude of the electron wave. The acceptable values of the total energy E of wave particle given by a solution is called eigen value and the corresponding function Ψ which gives eigen value of the energy, i s called eigen function.
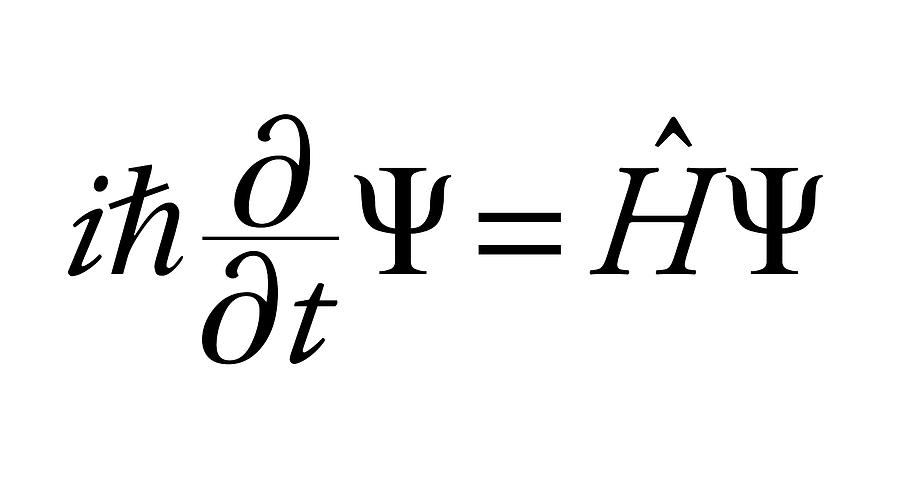
Out of these, only those solutions are significant which give certain definite and acceptable values of the total energy E. The Schrodinger wave equationis a second order differential equation.It has countless solutions., many of them having no significance.
